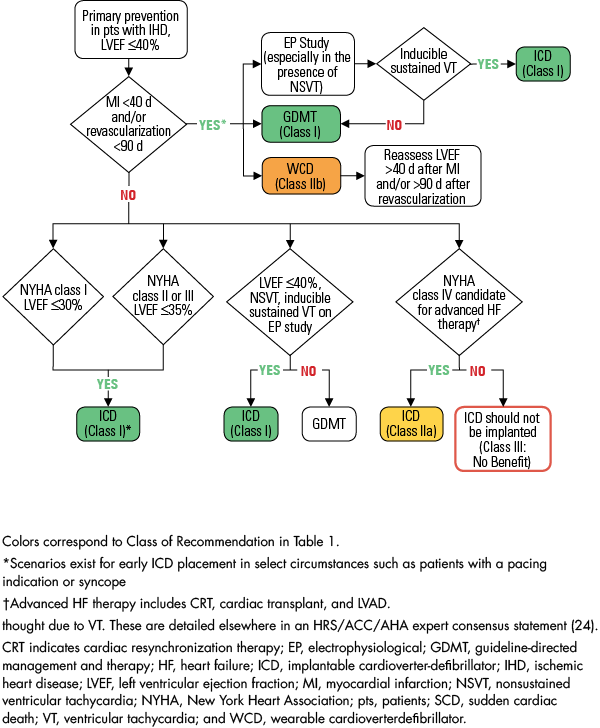
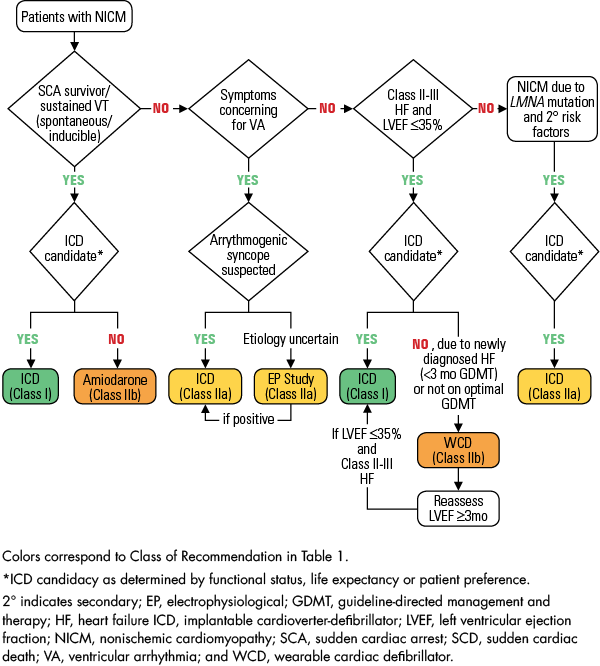
2017 AHA/ACC/HRS Guideline for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death | Circulation

Effectiveness, efficacy, and safety of wearable cardioverter-defibrillators in the treatment of sudden cardiac arrest – Results from a health technology assessment | International Journal of Technology Assessment in Health Care | Cambridge Core

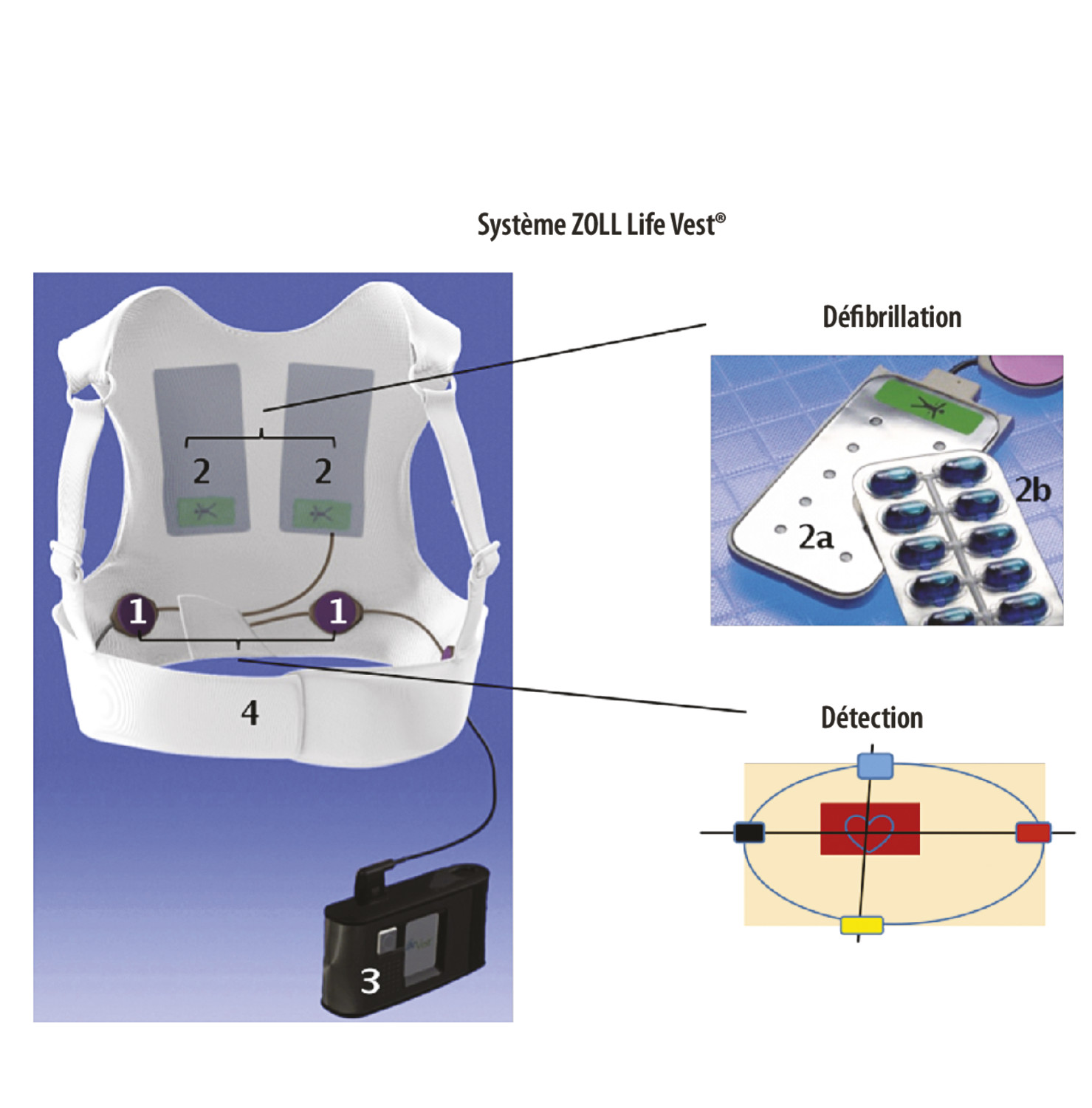
Sensors | Free Full-Text | Multiparameter Monitoring with a Wearable Cardioverter Defibrillator | HTML

The Wearable Cardioverter Defibrillator: A Life (Vest) of Controversy - American College of Cardiology