
Акустика Klipsch P-39F LH Merlo - купить в Киеве,доставка по Украине– цена,описание,характеристики,бесплатная доставка

Novel and Convenient Synthesis of Substituted Quinolines by Copper- or Palladium-Catalyzed Cyclodehydration of 1-(2-Aminoaryl)-2-yn-1-ols | The Journal of Organic Chemistry

Electrochemical Methods for Pd‐catalyzed C−H Functionalization - Erchinger - 2021 - Asian Journal of Organic Chemistry - Wiley Online Library

Green strategies for transition metal-catalyzed C–H activation in molecular syntheses - ScienceDirect

Computationally Assisted Mechanistic Investigation and Development of Pd-Catalyzed Asymmetric Suzuki–Miyaura and Negishi Cross-Coupling Reactions for Tetra-ortho-Substituted Biaryl Synthesis | ACS Catalysis

Novel and Convenient Synthesis of Substituted Quinolines by Copper- or Palladium-Catalyzed Cyclodehydration of 1-(2-Aminoaryl)-2-yn-1-ols | The Journal of Organic Chemistry

Electrochemical Methods for Pd‐catalyzed C−H Functionalization - Erchinger - 2021 - Asian Journal of Organic Chemistry - Wiley Online Library
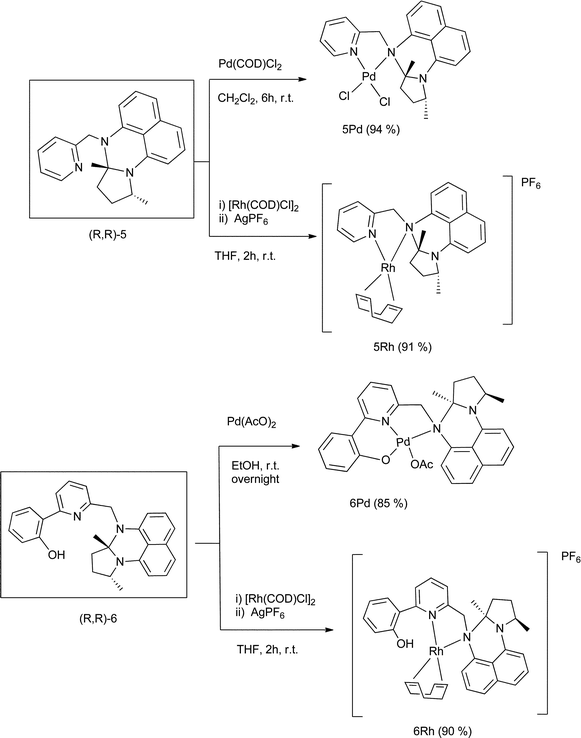
Development of homogeneous and heterogenized rhodium ( i ) and palladium(ii) complexes with ligands based on a chiral proton sponge building block and ... - Dalton Transactions (RSC Publishing) DOI:10.1039/C1DT10597C

Synthesis, Structural Characterization, and Catalytic Evaluation of Palladium Complexes with Homologous Ferrocene-Based Pyridylphosphine Ligands | Organometallics
Carbon‐Based Material‐Supported Palladium Nanocatalysts in Coupling Reactions: Discussion on their Stability and Heterogenei
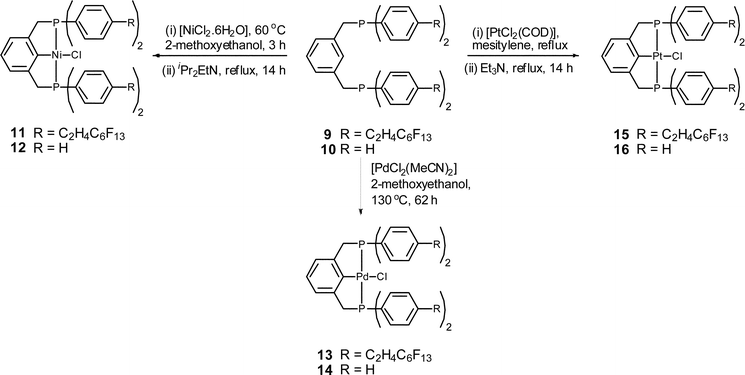
A recyclable perfluoroalkylated PCP pincer palladium complex - Dalton Transactions (RSC Publishing) DOI:10.1039/C0DT01004A

1,1'-P/O-Ferrocenyl Ligands in Palladium-Catalyzed Suzuki Coupling of Aryl Chlorides | Organometallics






